Window of opportunity in axial spondyloarthritis: A Stitch in Time
Abstract
Axial spondyloarthritis comprising both non-radiographic axial spondyloarthritis and ankylosingspondylitis has a deleterious impact on the patient’s quality of life with a detrimental outcome ofstructural damage. Although in the current era of diagnostic advancements, axSpA can be diagnosedearly within a short period after the onset of symptoms, but still there is a delay of up to severalyears in many parts of the world. The concept of a window of opportunity is primarily derived fromrheumatoid arthritis, which is relevant in the context of axSpA based upon the early diagnosis and tocommence highly effective treatment with biologics like anti-TNF and anti-IL-17 to modify thedisease process for arresting structural damage or syndesmophytes formation. Still, challenges existfor early diagnosis of SpA in patients with low back pain which ultimately creates a barrier toeffective treatment initiation. More robust researches along with the available evidence on both theaspects of clinical and imaging factors are the way forward for the early identification of susceptibleindividuals for early intervention with a better outcome.
Downloads
References
1. Lim, C.S.E., R. Sengupta, and K. Gaffney, The clinical utility of human leucocyte antigen B27 in axial spondyloarthritis. Rheumatology, 2018. 57(6): p. 959-968.
2. Siebert, S., R. Sengupta, and A. Tsoukas, Axial Spondyloarthritis. 2016: Oxford University Press.
3. Ward, M.M., et al., 2019 Update of the American College of Rheumatology/Spondylitis Association of America/Spondyloarthritis Research and Treatment Network recommendations for the treatment of ankylosing spondylitis and nonradiographic axial spondyloarthritis. Arthritis care & research, 2019. 71(10): p. 1285-1299.
4. Wendling, D., New targeted therapies in spondyloarthritis: what are the limits? 2019, Future Medicine.
5. Robinson, P.C. and M.A. Brown, The window of opportunity: a relevant concept for axial spondyloarthritis. Arthritis research & therapy, 2014. 16(3): p. 1-3.
6. Maksymowych, W.P., et al., Comparative effectiveness of secukinumab and adalimumab in ankylosing spondylitis as assessed by matching-adjusted indirect comparison: an analysis based on all pivotal phase 3 clinical trial data. Value in Health, 2017. 20(9): p. A935.
7. Betts, K., et al., OP0115 Relative Efficacy of Adalimumab versus Secukinumab in Active Ankylosing Spondylitis: A Matching-Adjusted Indirect Comparison. 2016, BMJ Publishing Group Ltd.
8. Fragoulis, G.E. and S. Siebert, Treatment strategies in axial spondyloarthritis: what, when and how? Rheumatology, 2020. 59(Supplement_4): p. iv79-iv89.
9. Mandl, P., et al., EULAR recommendations for the use of imaging in the diagnosis and management of spondyloarthritis in clinical practice. Annals of the rheumatic diseases, 2015. 74(7): p. 1327-1339.
10. Khmelinskii, N., A. Regel, and X. Baraliakos, The role of imaging in diagnosing axial spondyloarthritis. Frontiers in medicine, 2018. 5: p. 106.
11. Hermann, K.-G.A., et al., Descriptions of spinal MRI lesions and definition of a positive MRI of the spine in axial spondyloarthritis: a consensual approach by the ASAS/OMERACT MRI study group. Annals of the rheumatic diseases, 2012. 71(8): p. 1278-1288.
12. Yi, E., et al., Clinical, economic, and humanistic burden associated with delayed diagnosis of axial spondyloarthritis: a systematic review. Rheumatology and therapy, 2020. 7(1): p. 65-87.
13. Danve, A. and A. Deodhar, Axial spondyloarthritis in the USA: diagnostic challenges and missed opportunities. Clinical rheumatology, 2019. 38(3): p. 625-634.
14. Zhao, S.S., et al., Diagnostic delay in axial spondyloarthritis: a systematic review and meta-analysis. Rheumatology, 2021. 60(4): p. 1620-1628.
15. Hammoudeh, M., et al., Challenges of diagnosis and management of axial spondyloarthritis in North Africa and the Middle East: an expert consensus. Journal of International Medical Research, 2016. 44(2): p. 216-230.
16. Rusman, T., R.E. van Bentum, and I.E. Van der Horst-Bruinsma, Sex and gender differences in axial spondyloarthritis: myths and truths. Rheumatology, 2020. 59(Supplement_4): p. iv38-iv46.
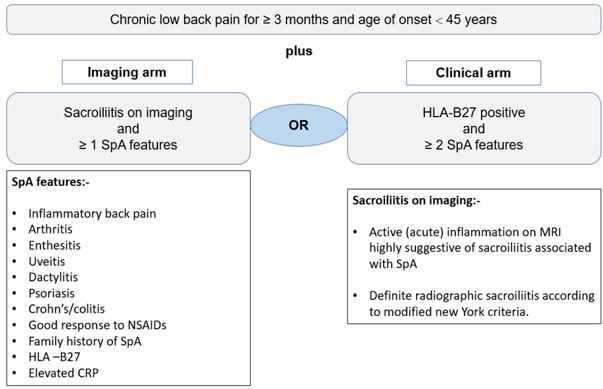
17. Rudwaleit, M., et al., The development of Assessment of SpondyloArthritis international Society classification criteria for axial spondyloarthritis (part II): validation and final selection. Annals of the rheumatic diseases, 2009. 68(6): p. 777-783.
18. de Winter, J., et al., Magnetic resonance imaging of the sacroiliac joints indicating sacroiliitis according to the Assessment of SpondyloArthritis international Society definition in healthy individuals, runners, and women with postpartum back pain. Arthritis & rheumatology, 2018. 70(7): p. 1042-1048.
19. Poddubnyy, D. and J. Sieper, Treatment of axial spondyloarthritis: what does the future hold? Current Rheumatology Reports, 2020. 22(9): p. 1-8.
20. van den Berg, R., et al., Classification of axial SpA based on positive imaging (radiographs and/or MRI of the sacroiliac joints) by local rheumatologists or radiologists versus central trained readers in the DESIR cohort. Annals of the rheumatic diseases, 2015. 74(11): p. 2016-2021.
21. Van Der Heijde, D., et al., 2016 update of the ASAS-EULAR management recommendations for axial spondyloarthritis. Annals of the rheumatic diseases, 2017. 76(6): p. 978-991.
22. Manica, S.R., et al., Effectiveness of switching between TNF inhibitors in patients with axial spondyloarthritis: is the reason to switch relevant? Arthritis research & therapy, 2020. 22(1): p. 1-6.
23. Garcês, S., J. Demengeot, and E. Benito-Garcia, The immunogenicity of anti-TNF therapy in immune-mediated inflammatory diseases: a systematic review of the literature with a meta-analysis. Annals of the rheumatic diseases, 2013. 72(12): p. 1947-1955.
24. Zhang, Z., et al., Risk of tuberculosis in patients treated with TNF-α antagonists: a systematic review and meta-analysis of randomised controlled trials. BMJ open, 2017. 7(3): p. e012567.
25. Elewski, B.E., et al., Association of secukinumab treatment with tuberculosis reactivation in patients with psoriasis, psoriatic arthritis, or ankylosing spondylitis. JAMA dermatology, 2021. 157(1): p. 43-51.
26. Baraliakos, X., et al., Comparison of the effects of secukinumab and adalimumab biosimilar on radiographic progression in patients with ankylosing spondylitis: design of a randomized, phase IIIb study (SURPASS). Clinical drug investigation, 2020. 40(3): p. 269-278.
27. Emery, P., et al., Cost effectiveness of secukinumab for the treatment of active ankylosing spondylitis in the UK. Pharmacoeconomics, 2018. 36(8): p. 1015-1027.
28. Rodriguez, V.R. and D. Poddubnyy, Old and new treatment targets in axial spondyloarthritis. RMD open, 2015. 1(Suppl 1): p. e000054.
29. Maksymowych, W.P., et al., Suppression of inflammation and effects on new bone formation in ankylosing spondylitis: evidence for a window of opportunity in disease modification. Annals of the rheumatic diseases, 2013. 72(1): p. 23-28.
30. Maksymowych, W.P., Biomarkers for diagnosis of axial spondyloarthritis, disease activity, prognosis, and prediction of response to therapy. Frontiers in immunology, 2019. 10: p. 305.
31. Maksymowych, W., et al., Fat metaplasia on MRI of the sacroiliac joints increases the propensity for disease progression in the spine of patients with spondyloarthritis. RMD open, 2017. 3(1): p. e000399.
32. Protopopov, M., et al., Radiographic sacroiliitis progression in axial spondyloarthritis: central reading of 5 year follow-up data from the Assessment of SpondyloArthritis international Society cohort. Rheumatology, 2021. 60(5): p. 2478-2480.
33. Dougados, M., et al., Sacroiliac radiographic progression in recent onset axial spondyloarthritis: the 5-year data of the DESIR cohort. Annals of the rheumatic diseases, 2017. 76(11): p. 1823-1828.
34. Huang, Y., et al., Impact of tumor necrosis factor α inhibitors on MRI inflammation in axial spondyloarthritis assessed by Spondyloarthritis Research Consortium Canada score: A meta-analysis. PloS One, 2020. 15(12): p. e0244788.
35. van der Heijde, D., et al., Limited radiographic progression and sustained reductions in MRI inflammation in patients with axial spondyloarthritis: 4-year imaging outcomes from the RAPID-axSpA phase III randomised trial. Annals of the rheumatic diseases, 2018. 77(5): p. 699-705.
36. Braun, J., et al., Secukinumab shows sustained efficacy and low structural progression in ankylosing spondylitis: 4-year results from the MEASURE 1 study. Rheumatology, 2019. 58(5): p. 859-868.
37. Maksymowych, W.P., et al., Clinical and MRI responses to etanercept in early non-radiographic axial spondyloarthritis: 48-week results from the EMBARK study. Annals of the rheumatic diseases, 2016. 75(7): p. 1328-1335.
38. Lorenzin, M., et al., An update on serum biomarkers to assess axial spondyloarthritis and to guide treatment decision. Therapeutic Advances in Musculoskeletal Disease, 2020. 12: p. 1759720X20934277.
39. Sieper, J., et al., Efficacy and safety of adalimumab in patients with non-radiographic axial spondyloarthritis: results of a randomised placebo-controlled trial (ABILITY-1). Annals of the rheumatic diseases, 2013. 72(6): p. 815-822.
40. Sieper, J., et al., A randomized, double‐blind, placebo‐controlled, sixteen‐week study of subcutaneous golimumab in patients with active nonradiographic axial spondyloarthritis. Arthritis & rheumatology, 2015. 67(10): p. 2702-2712.
41. Dougados, M., et al., Symptomatic efficacy of etanercept and its effects on objective signs of inflammation in early nonradiographic axial spondyloarthritis: a multicenter, randomized, double‐blind, placebo‐controlled trial. Arthritis & rheumatology, 2014. 66(8): p. 2091-2102.
42. Sieper, J., et al., Predictors of remission in patients with non-radiographic axial spondyloarthritis receiving open-label adalimumab in the ABILITY-3 study. RMD open, 2019. 5(1): p. e000917.
43. Dougados, M., et al., Efficacy and safety of ixekizumab through 52 weeks in two phase 3, randomised, controlled clinical trials in patients with active radiographic axial spondyloarthritis (COAST-V and COAST-W). Annals of the rheumatic diseases, 2020. 79(2): p. 176-185.
44. Deodhar, A., et al., Ixekizumab for patients with non-radiographic axial spondyloarthritis (COAST-X): a randomised, placebo-controlled trial. The Lancet, 2020. 395(10217): p. 53-64.
45. Deodhar, A., et al., Improvement of Signs and Symptoms of Nonradiographic Axial Spondyloarthritis in Patients Treated With Secukinumab: Primary Results of a Randomized, Placebo‐Controlled Phase III Study. Arthritis & Rheumatology, 2021. 73(1): p. 110-120.
46. San Koo, B. and T.-H. Kim, The role of ixekizumab in non-radiographic axial spondyloarthritis. Therapeutic Advances in Musculoskeletal Disease, 2021. 13: p. 1759720X20986734.
47. Magrey, M.N. and M.A. Khan, The paradox of bone formation and bone loss in ankylosing spondylitis: evolving new concepts of bone formation and future trends in management. Current rheumatology reports, 2017. 19(4): p. 17.
48. Haroon, N., et al., The impact of tumor necrosis factor α inhibitors on radiographic progression in ankylosing spondylitis. Arthritis & Rheumatism, 2013. 65(10): p. 2645-2654.
49. Baraliakos, X., et al., Long-term effects of secukinumab on MRI findings in relation to clinical efficacy in subjects with active ankylosing spondylitis: an observational study. Annals of the rheumatic diseases, 2016. 75(2): p. 408-412.
50. Jones, A., et al., Biologics for treating axial spondyloarthritis. Expert opinion on biological therapy, 2018. 18(6): p. 641-652.
51. Baraliakos, X., et al., Continuous long-term anti-TNF therapy does not lead to an increase in the rate of new bone formation over 8 years in patients with ankylosing spondylitis. Annals of the rheumatic diseases, 2014. 73(4): p. 710-715.
52. Molnar, C., et al., TNF blockers inhibit spinal radiographic progression in ankylosing spondylitis by reducing disease activity: results from the Swiss Clinical Quality Management cohort. Annals of the rheumatic diseases, 2018. 77(1): p. 63-69.
53. Braun, J., et al., The effect of two golimumab doses on radiographic progression in ankylosing spondylitis: results through 4 years of the GO-RAISE trial. Annals of the rheumatic diseases, 2014. 73(6): p. 1107-1113.
54. Jeong, H., et al., Effect of tumor necrosis factor α inhibitors on spinal radiographic progression in patients with ankylosing spondylitis. International journal of rheumatic diseases, 2018. 21(5): p. 1098-1105.
55. Braun, J., et al., Effect of secukinumab on clinical and radiographic outcomes in ankylosing spondylitis: 2-year results from the randomised phase III MEASURE 1 study. Annals of the rheumatic diseases, 2017. 76(6): p. 1070-1077.
56. Poddubnyy, D., et al., Rapid improvement in spinal pain in patients with axial spondyloarthritis treated with secukinumab: primary results from a randomized controlled phase-IIIb trial. Therapeutic advances in musculoskeletal disease, 2021. 13: p. 1759720X211051471.
57. Park, J.W., et al., Impact of tumor necrosis factor inhibitor versus nonsteroidal antiinflammatory drug treatment on radiographic progression in early ankylosing spondylitis: its relationship to inflammation control during treatment. Arthritis & Rheumatology, 2019. 71(1): p. 82-90.
58. Sieper, J., et al., Efficacy and safety of infliximab plus naproxen versus naproxen alone in patients with early, active axial spondyloarthritis: results from the double-blind, placebo-controlled INFAST study, Part 1. Annals of the rheumatic diseases, 2014. 73(1): p. 101-107.
59. Shimabuco, A.Y., et al., Factors associated with ASDAS remission in a long-term study of ankylosing spondylitis patients under tumor necrosis factor inhibitors. Advances in Rheumatology, 2019. 58.
60. Ziade, N.R. and X. Baraliakos, Tapering bDMARDs in axial SpA—what is the current evidence? Nature Reviews Rheumatology, 2019. 15(6): p. 322-324.
61. Li, K.-P., et al., Full dose, half dose, or discontinuation of etanercept biosimilar in early axial spondyloarthritis patients: a real-world study in China. Archives of medical science: AMS, 2019. 15(3): p. 700.
62. Gratacós, J., et al., Non-inferiority of dose reduction versus standard dosing of TNF-inhibitors in axial spondyloarthritis. Arthritis research & therapy, 2019. 21(1): p. 1-10.
63. Dubash, S., D. McGonagle, and H. Marzo-Ortega, New advances in the understanding and treatment of axial spondyloarthritis: from chance to choice. Therapeutic advances in chronic disease, 2018. 9(3): p. 77-87.
Copyright (c) 2022 Author (s). Published by Siddharth Health Research and Social Welfare Society

This work is licensed under a Creative Commons Attribution 4.0 International License.


 OAI - Open Archives Initiative
OAI - Open Archives Initiative


