Serum ferritin, serum LDH and d-dimer in correlation with the outcome in Covid-19.
Abstract
BACKGROUND: Early detection of comorbidities, severe patients with SARS-COV-2 and helpful diagnostic tools is very important for individual treatment and helps us in assessing the outcome of moderate to severe patients.
METHOD: In our study we have included 108 patients with RT-PCR positive for SARS COV-2 and the samples for D-dimer, Sr. LDH and Sr.ferritin were sent and analyzed.
RESULTS: the effect of D-dimer, serum LDH and serum ferritin individually and combinedly showed in our study there was no significant difference between D-dimer and the outcome however there was a significant p-value of <0.001 with serum LDH and the outcome, and p-value of 0.016 with serum ferritin and outcome.
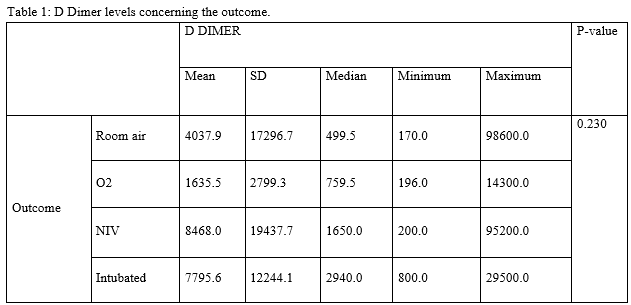
CONCLUSIONS: When the markers individually analyzed they showed a significant association between outcome and markers, in elevated O2and normal patients. Mean D dimer, serum ferritin and serum LDH were high among subjects who required NIV and low among subjects who required O2, on Room air and intubated respectively. In hospitalized patients with respiratory distress, we recommend clinicians closely monitor serum ferritin, serum LDH and D-dimer and also other markers of severity, WBC count, lymphocyte count, platelet count, IL-6 as markers for potential progression to critical illness, has been under the study, however our study sample being 108, a study with a larger number of samples required to predict the outcome.
Downloads
References
Cohen J. COVID-19 shot protects monkeys. Science. 2020;368:456–457.
Yuan M., Wu N.C., Zhu X., Lee C.-C.D., So R.T.Y., Lv H., Mok C.K.P., Wilson I.A. A highly conserved cryptic epitope in the receptor-binding domains of SARS-CoV-2 and SARS-CoV. Science. 2020:eabb7269.
Chen Y., Lu S., Jia H., Deng Y., Zhou J., Huang B., Yu Y., Lan J., Wang W., Lou Y. A novel neutralizing monoclonal antibody targeting the N-terminal domain of the MERS-CoV spike protein. Emerging Microbes and Infections. 2017;6:e37.
Ai T, Yang Z, Hou H, Zhan C, Chen C, Lv W, et al. Correlation of chest CT and RT-PCR testing in coronavirus disease 2019 (COVID-19) in China: A report of 1014 cases. Radiology 2020; 296 : E32-40
Fang Y, Zhang H, Xie J, Lin M, Ying L, Pang P, et al. Sensitivity of chest CT for COVID-19: Comparison to RT-PCR. Radiology 2020; 296 : E115-7.
Wang Y, Kang H, Liu X, Tong Z. Combination of RT-qPCR testing and clinical features for diagnosis of COVID-19 facilitates management of SARS-CoV-2 outbreak. J Med Viral 2020; 92 : 538-9.
Korber B, Fischer WM, Gnanakaran S, et al., Sheffield COVID-19 Genomics Group
. Tracking changes in SARS-CoV-2 spike: evidence that D614G increases infectivity of the covid-19 virus. Cell2020;182:812-827.e19. doi:10.1016/j.cell.2020.06.043 pmid:32697968
Monteil V,Kwon H,Prado P, et al,Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2. Cell2020;181:905-913.e7. doi:10.1016/j.cell.2020.04.004 pmid:32333836
Allegra A., Di Gioacchino M., Tonacci A., Musolino C., Gangemi S. Immunopathology of SARS-CoV-2 infection: immune cells and mediators, prognostic factors, and immune-therapeutic implications. Int. J. Mol. Sci. 2020;21:E4782.
Lega S., Naviglio S., Volpi S., Tommasini A. Recent insight into SARS-CoV2 immunopathology and rationale for potential treatment and preventive strategies in COVID-19. Vaccines (Basel) 2020;8:E224.
Stebbing J, Phelan A, Griffin I, et al. COVID-19: combining antiviral and anti-inflammatory
treatments. Lancet Infect Dis. 2020;20(4):400–2.
Fillebeen C, Wilkinson N, Charlebois E, et al. Hepcidin-mediated hypoferremic response to acute inflammation requires a threshold of Bmp6/Hjv/Smad signaling. Blood. 2018;132(17):1829–41.
Shoenfeld Y. Corona (COVID-19) time musings: our involvement in COVID-19 pathogenesis, diagnosis, treatment and vaccine planning. Autoimmun Rev. 2020;102538
Rosário C., Zandman-Goddard G., Meyron-Holtz E.G., D’Cruz D.P., Shoenfeld Y. The hyperferritinemic syndrome: macrophage activation syndrome, Still’s disease, septic shock and catastrophic antiphospholipid syndrome. BMC Med. 2013;11:185.
Kuhn, L.C. , 2015. Iron regulatory proteins and their role in controlling iron metabolism. Metallomics , 7, 232–243.
.Kernan, K.F. and Carcillo, J.A. , 2017. Hyperferritinemia and inflammation. International immunology , 29 (9), 401–409.
Kell, D.B. and Pretorius, E. , 2014. Serum ferritin is an important inflammatory disease marker, as it is mainly a leakage product from damaged cells. Metallomics: integrated biometal science , 6 (4), 748–773.
Han Y, Zhang H, Mu S, et al. Lactate dehydrogenase, a risk factor of severe COVID-19 patients. medRxiv 2020:2020.03.24.20040162. doi:10.1101/2020.03.24.20040162. Available at: https://www.medrxiv.org/content/10.1101/2020.03.24.20040162v1. Accessed 1 May 2020.
Kuang Z-S, Yang Y-l, Wei W, et al. Clinical characteristics and prognosis of community-acquired pneumonia in autoimmune disease-induced immunocompromised host: a retrospective observational study. World J Emerg Med 2020; 11:145–51.
Ding J, Karp JE, Emadi A. Elevated lactate dehydrogenase (LDH) can be a marker of immune suppression in cancer: Interplay between hematologic and solid neoplastic clones and their microenvironments. Cancer Biomark 2017; 19:353–63.
Mangalmurti N, Hunter CA. Cytokine Storms: understanding COVID-19. Immunity2020;53:1925. doi:10.1016/j.immuni.2020.06.017 pmid:32610079
Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med2020;180:934-43. doi:10.1001/jamainternmed.2020.0994 pmid:32167524
Huang, C. , et al. , 2020. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The lancet , 395 (10223), 497–506.
Consider cytokine storm syndromes and immunosuppression. Lancet , 395 (10229), 1033–1034. doi:10.1016/S0140-6736(20)30628-0.
Becker RC. COVID-19 update: Covid-19-associated coagulopathy. J Thromb Thrombolysis 2020; 50: 54–67
Smith SA, Mutch NJ, Baskar D, Rohloff P, Docampo R, Morrissey JH. Polyphosphate modulates blood coagulation and fibrinolysis. Proc Natl Acad Sci USA. 2006;103(4):903–908.
Subramaniam S, Jurk K, Hobohm L, et al. Distinct contributions of complement factors to platelet activation and fibrin formation in venous thrombus development. Blood. 2017; 129(16):2291-2302
Iba T, Levy JH. Inflammation and thrombosis: roles of neutrophils, platelets and endothelial cells and their interactions in thrombus formation during sepsis. J Thromb Haemost. 2018;16(2):231–241.
Noubouossie DF, Reeves BN, Strahl BD, Key NS. Neutrophils: back in the thrombosis spotlight. Blood. 2019;133(20):2186–2197.
Iba T, Levy JH, Wada H, Thachil J, Warkentin TE, Levi M; Subcommittee on Disseminated Intravascular Coagulation. Differential diagnoses for sepsis-induced disseminated intravascular coagulation: communication from the SSC of the ISTH. J Thromb Haemost. 2019; 17(2):415-419.
Levi M, Scully M. How I treat disseminated intravascular coagulation. Blood. 2018;131(8): 845-854.
Lippi G, Bonfanti L, Saccenti C, Cervellin G. Causes of elevated D-dimer in patients admitted to a large urban emergency department. Eur J Intern Med. 2014;25(1):45-48.
Connors JM, Levy JH. COVID-19 and its implications for thrombosis and anticoagulation. Blood 2020; 135: 2033–2040.
Copyright (c) 2021 Author (s). Published by Siddharth Health Research and Social Welfare Society

This work is licensed under a Creative Commons Attribution 4.0 International License.


 OAI - Open Archives Initiative
OAI - Open Archives Initiative


